Natural Herbal Prostate Support for Men 2
If you’re a man in your 40s or older who suffers with occasional urinary urgency, you’ve no doubt become increasingly aware of your prostate. And even if you haven’t personally experienced any prostate issues, you probably know other men who have. If you watch TV, especially sports broadcasts, it’s hard to miss all the drug commercials populated with middle-aged men doing fun, “manly” things, only to be interrupted for an emergency trip to the “john” at the 14th tee or making an embarrassed dash up crowded grandstand stairs in a packed stadium at a key moment in the game.
Benign Prostatic Hyperplasia
As men age, prostate enlargement – or Benign Prostatic Hyperplasia (BPH) – tends to become an increasingly common issue. And while BPH is a “benign” condition, it nevertheless can lead to serious discomfort. As men enter their 40s, their physician will usually recommend two common tests to evaluate prostate health: the dreaded digital rectal exam (DRE), and a blood test for a substance called prostate-specific androgen (PSA). PSA is an important biomarker that can indicate a potential problem with excess prostate growth. And even if you are symptom-free, these tests may alert your physician to early prostate enlargement.
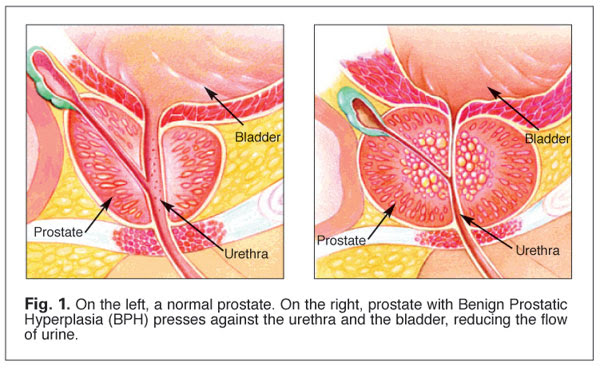
To appreciate the significance of an enlarged prostate it’s important to understand the basic anatomy of the male urinary tract. As urine forms in the kidneys it flows into the bladder via the ureters. As the bladder fills, its muscular walls begin to contract to force the urine through the urethra to pass out of the body.
Sitting just below the bladder, the prostate gland completely wraps itself around the urethra. Normally about the size of a walnut, all is well until the prostate begins to enlarge. As it enlarges, the prostate begins to compress the urethra, blocking normal urine flow and often turning what was once a strong stream into a mere trickle (Fig. 1). In response to the increasing resistance to urine flow, the bladder walls can become irritated and begin to thicken. This results in the bladder contracting, even when it contains very little urine, resulting in a sudden and urgent need to urinate. In time, the growing need for frequent, urgent trips to the bathroom can become increasingly frustrating, especially when trying to get a good night’s sleep – or finish a round a golf.
Over time, the bladder muscle can become so weak that it can lose its ability to completely empty itself. As a result, the residual urine remaining in the bladder can act as a reservoir that harbors bacterial growth, leading to urinary tract infections (UTIs) and more seriously, potential kidney infections.
Incidence of Prostate Enlargement
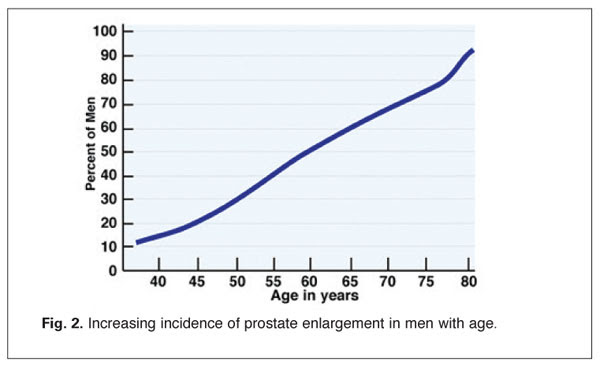
Prostate enlargement affects an estimated 60 percent of men between the ages of 40 and 60 years, and up to 75 percent of men over age 60 (Fig. 2).(1) BPH symptoms become clinically significant in about half of these men, accounting for at least 1.7 million doctor visits per year in the United States, with estimated annual health care costs exceeding $4 billion. In addition to the costs, symptoms of BPH have been shown to have a significant negative impact on patient-reported quality of life and psychological well-being.
Prostate enlargement may begin as early as one’s 20s, but such growth is usually slow and symptoms of significant growth don’t usually begin to appear until a man reaches middle age or older. This has led some researchers to propose that BPH develops in two phases. In the first phase, which has no clinical signs, microscopic changes within the prostate may occur as early as the third or fourth decade of life.
The second phase, which typically begins in the fifth or sixth decade of life, is marked by macroscopic changes – i.e., prostate enlargement. However, clinical signs of the disorder manifest only if enlargement is substantial and becomes complicated by other disorders, such as prostatitis (inflammation of the prostate gland), or if the gland becomes hardened or deformed.
It has been suggested that, while nearly all men experience the microscopic changes in the prostate, if they live long enough, only about half will experience prostate enlargement, and, of those, only about half will develop clinical symptoms. The question then arises: What causes these relatively common prostate changes that lead to clinical disease?
Risk Factors for BPH
Epidemiologic studies have demonstrated that many of the risk factors associated with cardiovascular diseases are also associated with increased risk of BPH. These risk factors include obesity, hypertension, and diabetes. Age-related impairment of blood circulation to the prostate, caused by atherosclerosis, also plays a key role in the development of BPH. 1 And the connection between BPH and Type 2 diabetes is considered especially strong. 2-4 For example, excessive insulin levels, a primary sign of insulin resistance in Type 2 diabetes, may be a direct contributor to prostate growth.
Hormones and BPH
Hormones are known to affect the development and progression of BPH. This fact is illustrated by the unusual finding that men with cirrhosis of the liver have a lower incidence of BPH than men with normal liver function. Men with cirrhosis of the liver have been found to have a lower incidence of BPH than men with normal liver function, probably because the liver damage reduces the metabolism of hormones to compounds that adversely influence prostate cell growth.
Not all hormonal influences on BPH have been determined, but sex hormones (e.g., estrogen, testosterone, prolactin) and insulin have been shown to have such an effect.
In particular, it is thought that the conversion of testosterone to dihydrotestosterone (DHT) may be a significant risk factor for BPH.
Drugs like finasteride and dutasteride, which reduce symptoms of BPH, suppress DHT production by inhibiting the enzyme 5α-reductase, which converts testosterone to DHT. These drugs also reduce male-pattern balding, an effect that is likely related to the fact that DHT is produced in the sebaceous glands.
Recognizing Signs of BPH
As noted above, BPH is an extremely slow process, beginning in a man’s mid- to late 20s, but rarely showing clear symptoms until at least a quarter-century later. Symptoms tend to appear slowly and insidiously so that you probably won’t notice them until they’re pretty far along. Common symptoms of prostate enlargement include:
|
|
No one really understands why BPH occurs, although for centuries the condition has been associated with aging. Today we know that men’s bodies (primarily the testes) produce large amounts of testosterone (the “male hormone”) and smaller amounts of estrogen (the “female hormone”). With advancing age, active testosterone production gradually declines, resulting in a higher proportion of estrogen to testosterone. Animal studies suggest that BPH may occur because the higher ratio of estrogen to testosterone in the prostate increases the activity of substances that promote cell growth. 5
According to a second theory, BPH may result from the presence of excess DHT in the prostate. Some research suggests that even as blood testosterone levels decline, the prostates of some older men continue to produce high levels of DHT. Conversely, research also shows that men who do not produce DHT do not develop BPH. 5
A third possible cause for BPH is linked to “instructions” that prostate cells receive early in life. Cells in one section of the prostate are thought to follow these instructions and “reawaken” later in life; either delivering signals to other cells in the gland to make them grow, or making them more sensitive to hormones that influence growth. 5
Inflammation and BPH
Chronic inflammation is another factor shown to play an important role in the initiation of BPH and many other diseases. 6 Is inflammation a cause or a consequence of BPH? One analysis showed that bacterial and noninfectious chronic prostatitis could represent “inciting” factors leading to tissue hyperproliferation, possibly via the recently demonstrated antigen-presenting capacity of prostatic stromal cells, which enables them to induce and sustain intraglandular immune responses. 7
According to a major meta-analysis, chronic inflammation appears to be one of these predictive factors. The clinical studies showed a relationship between chronic prostatic inflammation and prostate volume or urinary symptoms. In microscopic studies, numerous inflammatory cells were found to be infiltrating BPH tissues.
Immune cells release cytokines and growth factors that modulate the immune response, but evidence also shows that they promote prostatic cell growth. In addition, prostatic cells themselves are able to secrete inflammatory mediators and stimulate their own growth. Once this vicious circle gets under way, it appears that feedback controls can be overwhelmed, leading to progressive increases in prostate volume. 8
In another study connecting inflammation with BPH, inflammatory cells were noted in 282 patients treated by surgery for a complicated and/or symptomatic BPH. Inflammatory cells were found to have infiltrated prostate tissues in a majority of the patients, indicating a role for inflammation in the development of BPH based on the incidence of inflammation, prostate volume, and other measures. These findings suggest that progressive prostate enlargement may be strongly influenced by chronic inflammatory processes. 9-10
Overall, recent evidence indicates that BPH is a complex disease, but one of the mechanisms leading to prostatic enlargement and urinary symptoms is chronic prostatic inflammation, thus suggesting that inflammation may be a therapeutic target for BPH.
Options for BPH
If you follow the recommendations of the National Institutes of Health (NIH), which are strongly influenced by the pharmaceutical industry, men with BPH have a limited range of choices for treating their condition, and none of them are terribly satisfactory.\
• “Watchful Waiting” involves close monitoring of prostate growth. If growth is minimal, no further treatment may be necessary.
• Surgery, the unkindest cut of all. Various forms of invasive and minimally invasive procedures, include open prostatectomy, transurethral resection of the prostate (TURP) laser surgery, transurethral microwave procedures, photoselective vaporization of the prostate (PVP), interstitial laser coagulation, transurethral needle ablation, water-induced thermotherapy, cryoablation, and high-intensity focused ultrasound (HIFU).
• Pharmaceutical Therapy. Several drugs have been approved by the FDA to treat BPH. In addition to finasteride and dutasteride, other drugs known as alpha-blockers (doxazosin, tamsulosin, alfuzosin and terazosin) have been approved to improve urine flow and reduce bladder outlet obstruction.
Traditional Herbs for BPH
Although some studies have shown as much as a 20 percent decrease in prostate volume in up to 50 percent of men treated with 5α-reductase inhibitors, as with most drugs, this approach can be expensive and prone to side effects, including sexual dysfunction. In order to avoid these side effects, millions of men have turned to alternatives, including phytonutrients such as Serenoa repens (Saw Palmetto Berry) and Pygeum africanum (African Plum). Indeed, early clinical studies indicated that saw palmetto berry extract was helpful for relieving symptoms of BPH, and by 2006 saw palmetto was the third-highest-selling herbal dietary supplement in the U.S., after garlic and echinacea.
Promising New Herbal Options
A growing body of modern research is beginning to help scientists better understand the underlying therapeutic mechanisms of traditional phytonutrients that have historically been used to support urinary function and prostate health. The following herbs are but a small selection of phytonutrients that have all been shown to be particularly effective for exerting beneficial effects on physiological systems (inflammation, excessive insulin, oxidative stress, etc.) that are known to contribute to prostate enlargement and BPH.
• Achyranthes Bidentata is a perennial herb used in traditional medicine to promote circulation, nourish the liver and kidneys, and support healthy urination. In China, Achyranthes is highly valued for its inflammation-modulating actions, and in Nepal, its root juice is commonly used for toothaches. Modern analytical research has revealed a variety of beneficial plant compounds in Achyranthes, including triterpenoid saponins, sitosterol, polysaccharides and alkaloids, that lend credence to its traditional uses. One important compound recently isolated from Achyranthes is oleanolic acid, a naturally occurring triterpenoid possessing liver-protective and immuno-supportive properties. 11-14
• Alpinia Oxyphylla, a medicinal plant closely related to ginger, is used in traditional Chinese formulas to control frequent urination and loss of bladder control. In Korea, where Alpinia also has a long history of use in support of healthy urination, researchers have found that Alpinia extracts protect brain cells (neurons) from cell death when the blood supply is restricted or cut off (ischemia). Alpinia was also shown to protect cells from damage caused by lipid peroxidation, a form of oxidative stress that occurs when free radicals attack and damage the lipids (fats) that comprise cell membranes. 15-16
• Cistanche Deserticola – The earliest description of the health benefits of the herb, Cistanches Deserticola, can be traced back almost 2,000 years, to the oldest known book on herbal medicine, the Shennong Bencao Jing, written in the year 100 A.D. Since then, Cistanches has become an important mainstay of traditional medicine in China and Japan, and today is a revered essence tonic recommended for the prevention and treatment of age-related diseases, for relieving fatigue, resolving infertility and supporting erectile function in men. 17-20
• Curcuma Zedoaria is a traditional herb closely related to turmeric (Curcuma longa) and curcuma (Curcuma aromatica). In addition being a powerful anti-inflammatory, with efficacy comparable to that of indomethacin (a non-steroidal anti-inflammatory drug commonly used to reduce fever, pain, stiffness, and swelling), 21 Zedoaria has also been shown to significantly inhibit production of Tumor necrosis factor-alpha (TNF-α), a major cytokine involved in systemic inflammation, and one of a group of cytokines involved in stimulating the acute phase reaction. 22
In 2008, Chinese researchers evaluated a popular traditional herbal formula containing the herbs listed above. After a treatment period lasting four weeks, the therapeutic efficacy of the formula was evaluated for improvements in frequency of night urination, degree of pain during urination, force of urinary flow, maximum flow rate, residual urine volume and prostate volume. The researchers noted that after only four weeks, residual urine volume was reduced, compared to before treatment. And while there were no obvious changes in prostate volume, the researchers found the formula to be “effective and safe for the treatment of benign prostatic hyperplasia.” 23
• Plantago Asiatica is one of the most popular folk herbal medicines in Asia. In 2008, Korean researchers demonstrated that a methanol extract (PAE) of Plantago possessed a potent antioxidant potential, comparable to that of vitamin C (ascorbate). Additionally, Plantago extract was shown to significantly inhibit formation of damaging Advanced Glycation Endproducts (AGEs) as effectively as the standard antiglycation agent, aminoguanidine. The glycation process involves non-enzymatic reactions between sugars and proteins that lead to the formation of these abnormal molecules that have been shown to play a pivotal role in the development of diabetic and aging-related health issues.24,25
• Nelumbo Nucifera, also known as Lotus, is a fresh-water plant that has been used as an anti-obesity agent in traditional Chinese herbal medicine for over 2,000 years. In 2009, scientists from the Research Institute of Chinese Medicine demonstrated that a compound in Nelumbo, called Neferine, enhances insulin sensitivity. In their paper the researchers compared the effects of neferine and rosiglitazone, an anti-diabetic drug in the thiazolidinedione class of drugs, on insulin-resistant rats. Their study found that levels of fasting blood glucose, fasting blood insulin, triglycerides and tumor necrosis factor-alpha decreased significantly in the rosiglitazone and neferine groups. Significantly, neferine was shown to be more effective at lowering plasma glucose levels than rosiglitazone.26
Summary
Each of these herbs have been shown to support prostate and urinary health by addressing a variety of biological mechanisms implicated in BPH: chronic inflammation, poor circulation, excess insulin levels, impaired liver function (leading to imbalanced hormone production), urine retention (resulting in bacterial infections), and altered immune responses known to induce tissue hyperproliferation.
While each of these herbs have been shown to support some aspect of urinary function and prostate health, the traditional approach of combining specific amounts and ratios of herbs into an advanced “essence” formula can led to superior results over those of single herbs.
References
1. Corder EH, Chute CG, Guess HA, Beard CM, O’Fallon WM, Lieber MM. Cancer Causes Control. May 1994;5(3):207-214.
2. Berger AP, Bartsch G, Deibl M, et al. BJU Int. Nov 2006;98(5):1038-1042.
3. Parsons JK, Carter HB, Partin AW, et al. J Clin Endocrinol Metab. Jul 2006;91(7):2562-2568.
4. Stamatiou K, Lardas M, Kostakos E, Koutsonasios V, Michail E. Adv Urol. 2009:818965.
5. National Institutes of Health. 2006. October 18, 2010.
6. Starsichova A, Lincova E, Pernicova Z, Kozubik A, Soucek K. Cell Signal. Nov 2010;22(11):1734-1744.
7. Fibbi B, Penna G, Morelli A, Adorini L, Maggi M. Int J Androl. Jun 1 2010;33(3):475-488.
8. Robert G, Salagierski M, Schalken JA, de La Taille A. Prog Urol. Jun 2010;20(6):402-407.
9. Robert G, Descazeaud A, Nicolaiew N, et al. Prostate. Dec 1 2009;69(16):1774-1780.
10. Bent S, Kane C, Shinohara K, et.al. N Engl J Med. 2006 Feb 9;354(6):557-66.
11. Liu J (1995). Journal of Ethnopharmacology 49(2):57–68.
12. Vetrichelvan, T and M. Jegadeesan. Indian Journal of Pharmacology 34, 2, 115-118. (2000b)
13. Lu T, Mao C, Zhang L, Xu W. Zhong Yao Cai. 1997 Oct;20(10):507-9.
14. Yu S, Zhang Y. Zhon ghua Zhong Liu Za Zhi. 1995 Jul;17(4):275-8.
15. Koo, B.S., Lee, W.C., Chang, Y.C., Kim, C.H. (2004). Phytotherapy Research, 18: 142–148.
16. Shi GF, An LJ, Jiang B, Guan S, Bao YM. Neurosci Lett. 2006 Aug 7;403(3):206-10. Epub 2006 Jun 23.
17. Lin LW, Hsieh MT, Tsai FH, Wang WH, Wu CR. J Ethnopharmacol. 2002 Dec;83(3):177-82.
18. Yang JH, Hu JP, Rena K, Du NS. Zhong Yao Cai. 2009 Jul;32(7):1067-9.
19. He WJ, Fang TH, Ma X, Zhang K, Ma ZZ, Tu PF. Planta Med. 2009 Oct;75(13):1400-4. Epub 2009 May 25.
20. Ko KM, Leung HY. Chin Med. 2007 Mar 27;2:3.
21. Makabe H, Maru N, Kuwabara A, Kamo T, Hirota M. Nat Prod Res. 2006 Jun;20(7):680-5.
22. Jang MK, Sohn DH, Ryu JH. Planta Med. 2001 Aug;67(6):550-2.
23. Guo J, Song CS, Han Q. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2008 Dec;28(12): 1082-5.
24. Choi SY, Jung SH, Lee HS, et.al. Phytother Res. 2008 Mar;22(3):323-9.
25. Huang DF, Tang YF, Nie SP, et.al. Eur J Pharmacol. 2009 Oct 12;620(1-3):105-11. Epub 2009 Aug 5.
26. Pan Y, Cai B, Wang K, et.al. J Ethnopharmacol. 2009 Jul 6;124(1):98-102. Epub 2009 Apr 11.


Hi there
… I live in Toronto Canada and want to buy your combining specific amounts and ratios of herbs into an advanced “essence formula” for Prostate you mention above …
Thanking for understanding, I am looking in advance to hear from you a positive reply.
Bardhyl Skenduli
Toronto
Hi Bardhyl,
I think you may be looking for https://www.tangoherbs.com/product/prostaphase-120-caps/